Your location: Home > News
Aleniglipron (CAS: 2685823-26-9) is an oral GLP-1 small molecule, just like Orforglipron, designed as a weight-loss API.
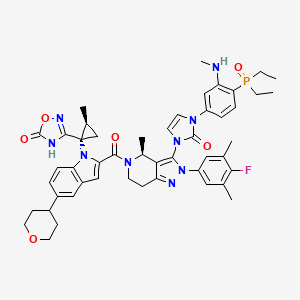
Recently, Structure Therapeutics announced the topline results of the Phase II ACCESS II trial for once-daily oral Aleniglipron, creating a global buzz in the weight-loss drug field.
Aleniglipron shows no plateau effect and significantly improved tolerability, earning multiple institutions’ “best-in-class” rating — positioning it as a strong challenger to Eli Lilly’s Orforglipron.

Key findings: after 44 weeks, Aleniglipron achieved up to 16.3% weight loss, compared to Orforglipron’s ~12.4% over 72 weeks.
In terms of efficacy, current data indicate Aleniglipron outperforms Orforglipron. Regarding safety and adherence: slow titration and high selectivity reduce gastrointestinal side effects and dropout rates, whereas Orforglipron shows higher GI adverse events and discontinuation rates.
Currently, Aleniglipron is in Phase II trials in the U.S., under clinical application in China, and in Phase I trials in Australia..jpg)
Hangzhou Royal Pharmaceutical Equipment Co., Ltd. specializes in pharmaceutical chemicals, APIs, and intermediates, with a focus on the B2B market for weight-loss drugs like Aleniglipron and Orforglipron, exporting to over 30 countries worldwide.

Website: https://www.jlpharms.com/
Tel: +86-571-85829053
Fax: +86-571-85829153
Address: Room 1702, Building 3, Greenland Central Plaza, 98 Daguan Road, Hangzhou, Zhejiang, China